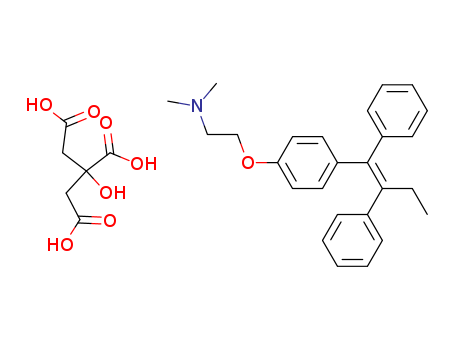
CasNo: 54965-24-1
Molecular Formula: C26H29NO.C6H8O7
Appearance: white to off-white powder
|
Application |
Tamoxifen, marketed under the brand name Nolvadex, has played a pivotal role in breast cancer management and prevention. Its long-term use has proven effective in reducing the recurrence rate of breast cancer, particularly in postmenopausal women with estrogen or progesterone receptor-positive tumors. The drug's risk-lowering effects persist for an extended period, with many of its side effects diminishing after the initial 5-year treatment period. Tamoxifen has been a mainstay in adjuvant therapy for breast cancer since the 1980s, benefiting millions of patients worldwide. It is administered in various doses and durations, depending on the region and specific clinical indications. Beyond its role in treating invasive breast cancer, Tamoxifen is also used to prevent invasive breast cancer in high-risk women and to reduce the risk of recurrence in women with ductal carcinoma in situ (DCIS). Additionally, it finds application in stimulating ovulation in some cases. With its widespread acceptance and efficacy, Tamoxifen has significantly contributed to improving the outcomes and quality of life for breast cancer patients and remains an essential tool in the fight against this prevalent and life-threatening disease. |
|
Chemical Properties |
white to off-white powder |
|
Originator |
Nolvadex,I.C.I.,UK,1973 |
|
Uses |
estrogen antagonist, antineoplastic |
|
Brand name |
Nolvadex (AstraZeneca); Soltamox (Savient). |
|
Therapeutic Function |
Antiestrogen, Antineoplastic |
|
Biochem/physiol Actions |
Cell permeable: yes |
InChI:InChI=1/C26H29NO.C6H8O7/c1-4-25(21-11-7-5-8-12-21)26(22-13-9-6-10-14-22)23-15-17-24(18-16-23)28-20-19-27(2)3;7-3(8)1-6(13,5(11)12)2-4(9)10/h5-18H,4,19-20H2,1-3H3;13H,1-2H2,(H,7,8)(H,9,10)(H,11,12)/p-3/b26-25-;
The cellular actions of tamoxifen are not completely understood, but it appears that the drug's antiproliferative effects are mediated primarily by inhibition of the activities of estrogen through binding to estrogen receptors (ERs).
The discovery that the simple hydrocarbons stilbene and triphenylethylene have weak estrogenic properties in the mouse stimulated studies of structure-activity relationships to develop …
A method for the treatment of emesis in ...
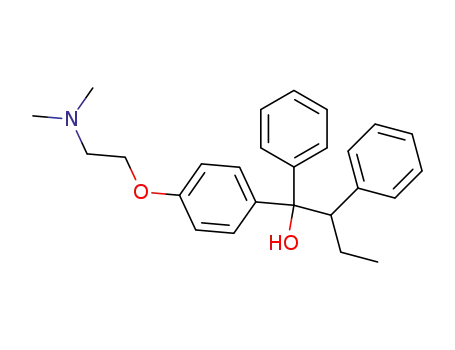
1-(4-(2-(dimethylamino)ethoxy)phenyl)-1,2-diphenylbutan-1-ol

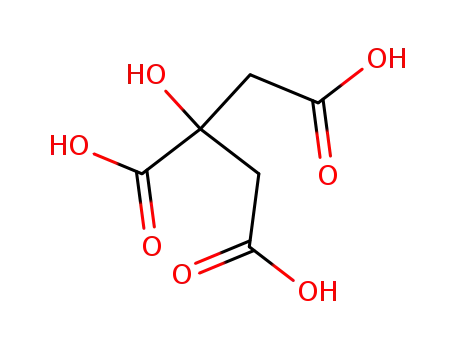
citric acid

tamoxifen citrate

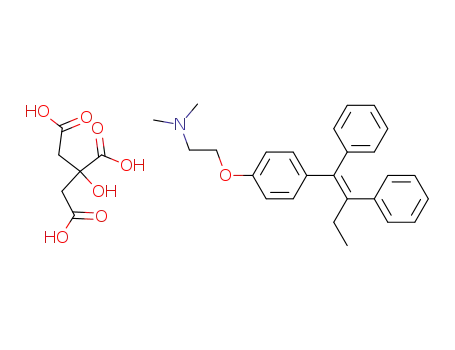
(Z)-2-(4-(1,2-diphenylbut-1-enyl)phenoxy)-N,N-dimethylethylamine citrate
| Conditions | Yield |
|---|---|
|
1-(4-(2-(dimethylamino)ethoxy)phenyl)-1,2-diphenylbutan-1-ol; With hydrogenchloride; In water; isopropyl alcohol; for 5h; Reflux;
citric acid; In acetone; at -5 - 10 ℃; for 10h;
|
7.5 g |

1-(4-(2-(dimethylamino)ethoxy)phenyl)-1,2-diphenylbutan-1-ol


citric acid


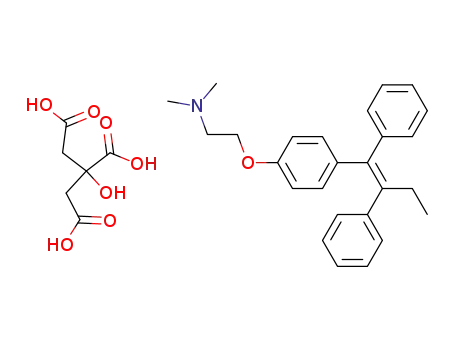
tamoxifen citrate


(Z)-2-(4-(1,2-diphenylbut-1-enyl)phenoxy)-N,N-dimethylethylamine citrate
| Conditions | Yield |
|---|---|
|
1-(4-(2-(dimethylamino)ethoxy)phenyl)-1,2-diphenylbutan-1-ol; With hydrogenchloride; In water; isopropyl alcohol; for 5h; Reflux;
citric acid; In acetone; at -5 - 10 ℃; for 10h;
|
7.5 g |
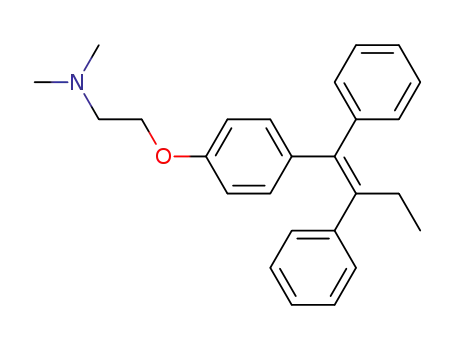
tamoxifen

citric acid

1-(4-(2-(dimethylamino)ethoxy)phenyl)-1,2-diphenylbutan-1-ol