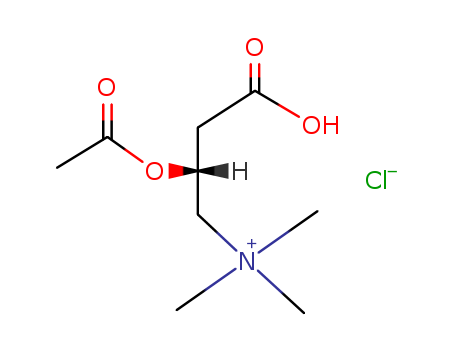
CasNo: 5080-50-2
Molecular Formula: C9H17NO4.HCl
Appearance: Crystallline powder
|
Chemical Properties |
Crystallline powder |
|
Originator |
Sigma-Tau (Italy) |
|
Uses |
Levacecarnine hydrochloride, structurally akin to the natural compound L-carnitine, serves as a nootropic agent with potential benefits in addressing cognitive disorders among the elderly, possibly attributed to its modest cholinergic properties. O-Acetyl-L-carnitine hydrochloride, on the other hand, acts as a cholinergic agonist, stimulating neuronal responses linked to serotonin and acetylcholine. This acetylated form of carnitine demonstrates antinociceptive properties, potentially mediated through heightened activity of muscarinic cholinergic receptors or mGlu2 glutamate receptors. Acetyl L-carnitine (ALC) plays a pivotal role in intermediary metabolism and has implications in enhancing female fertility by regulating the oxidative and metabolic aspects of the female reproductive system. ALC exhibits cholinomimetic qualities and possesses anti-inflammatory effects. It also modulates the γ-amino butyric acid (GABA) system and can influence the rate of glucose utilization within the brain, contributing to its neuroprotective function, particularly in the developing brain. |
|
Brand name |
NICETILE; BRANICEN |
InChI:InChI=1/C9H17NO4.ClH/c1-7(11)14-8(5-9(12)13)6-10(2,3)4;/h8H,5-6H2,1-4H3;1H/t8-;/m1./s1
Two C-2 monodeuterated isomers of l-carnitine were synthesized through two distinct enzymatic processes. The first involved the enzymatic hydration of crotonobetaine in D2O, while the second employed enzymatic proton exchange using l-[2-2H2] carnitine in H2O. These enzymatic reactions, catalyzed by an induced Escherichia coli carnitine hydrolyase, exhibited stereospecific behavior. Subsequently, we subjected the two isomers of l-[2-2H]carnitine to scrutiny via 1H NMR analysis at 500 MHz. This approach enabled us to independently track the pD-dependent variations and coupling constants of the H-2 protons.
On the other hand, the time spent in the target quadrant was increased significantly in animals treated with O-acetyl-L-carnitine/TE or nicotine/ O-acetyl-L-carnitine/TE compared to TE-…
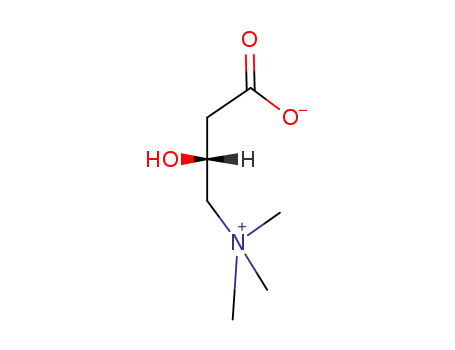
L-carnitine

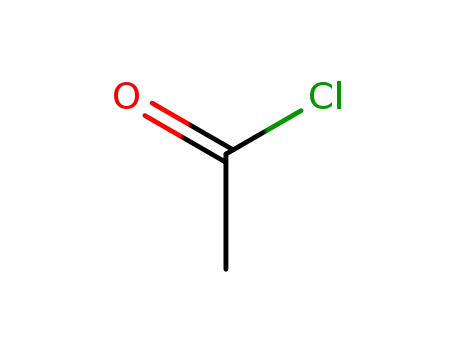
acetyl chloride

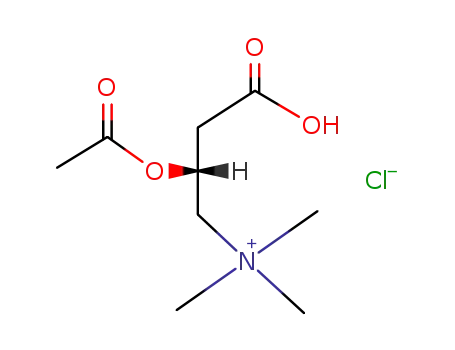
O-acetyl-L-carnitine hydrochloride
| Conditions | Yield |
|---|---|
|
With acetic acid; In Isopropyl acetate; at 10 - 60 ℃; for 8h; Solvent; Large scale;
|
86.3% |
|
In acetic acid; at 80 ℃; for 3h;
|
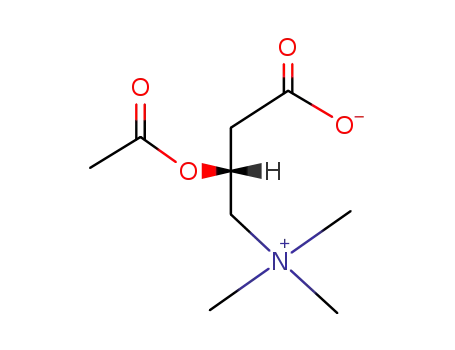
Acetyl-L-carnitine


O-acetyl-L-carnitine hydrochloride
| Conditions | Yield |
|---|---|
|
With hydrogenchloride; In water; Sealed tube; Darkness;
|
89% |
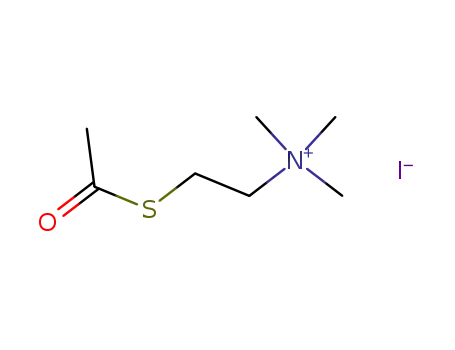
acetylthiocholine iodide
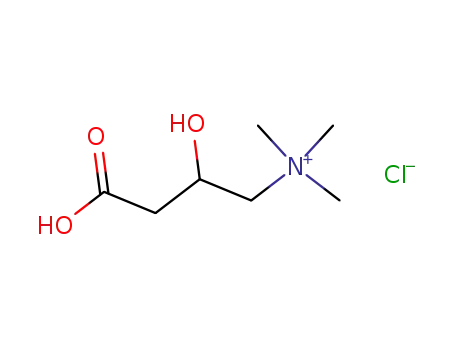
dl-carnitine hydrochloride
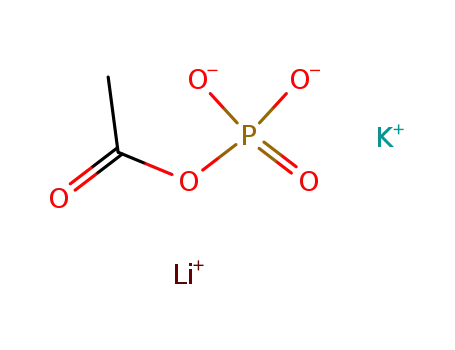
acetyl phosphate K+, Li+ salt

L-carnitine

Acetyl-L-carnitine